Now in the next step we have to check whether these s=o bonds are polar or nonpolar. These forces are stronger than forces between nonpolar molecules but less strong than ionic forces.

Covalent Bonds | Biology For Majors I
Positively charged h atoms make hydrogen bonds with negatively charged.
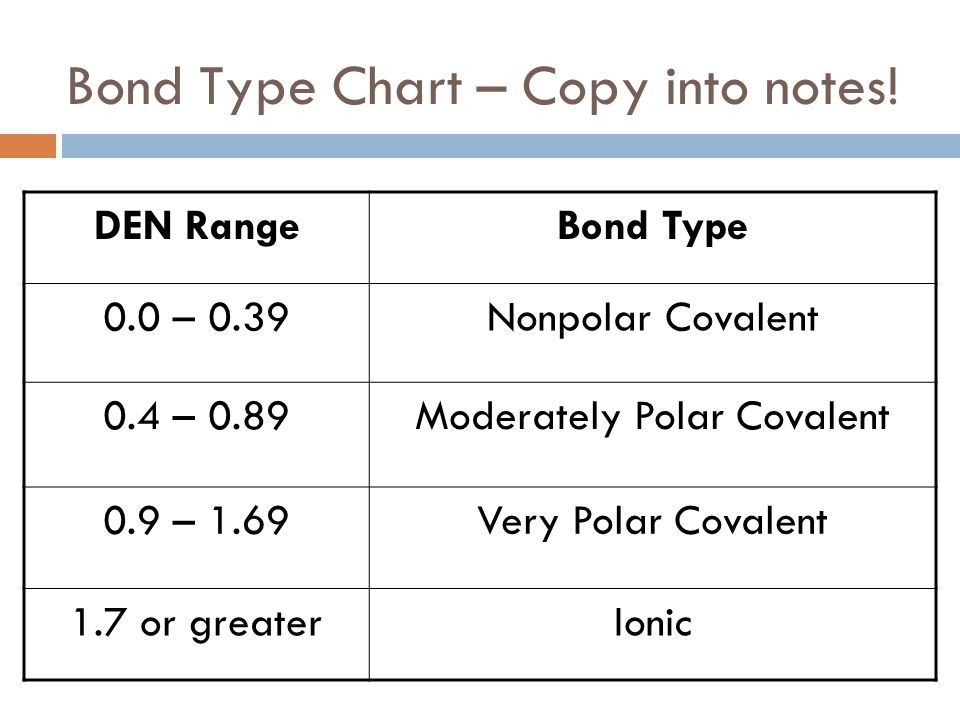
Polar nonpolar ionic. The general rule of thumb is that if the difference in electronegativity is greater. Now in the next step we have to check whether these bonds are polar or nonpolar. The reverse statement is that the values tend to decrease going down and to the left.
In chemistry, polarity is a separation of electric charge leading to a molecule or its chemical groups having an electric dipole moment, with a negatively charged end and a positively. Why does it matter if a molecule is polar or nonpolar? This organic chemistry video tutorial explains how to identify a bond as an ionic bond, polar covalent bond, or a nonpolar covalent bond.
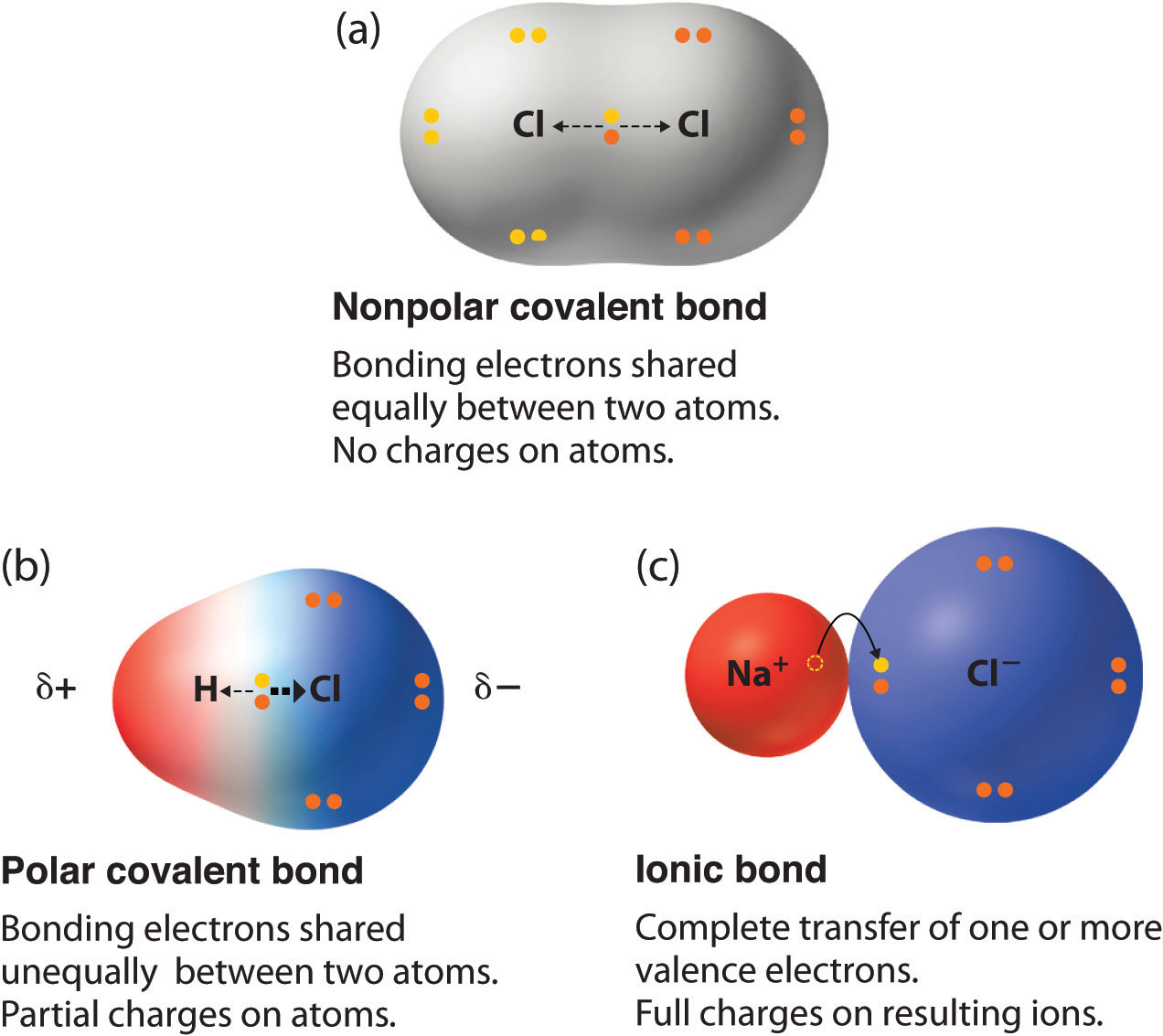
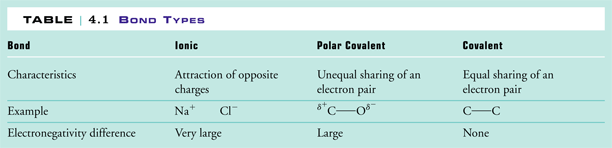
Ionic bonds are formed when there is very high difference of electronegativity in the elements. They have partial separation of charges. What dictates which kind of bond will form?
This pattern will help when you are asked to put several bonds in order from most to least ionic without. Polar molecules are the covalent molecules with partial ionic character. Ionic compounds are soluble in polar solvents and insoluble in nonpolar solvents.
And we also have to check the molecular geometry of so2. Polar, nonpolar, and ionic bonding information: There are two types of covalent bonding:
The kinetic traces of the. The polarity of a molecule is important because it. Polar covalent bond is formed when slight polarity is formed between the two elements due to.
And we also have to check the molecular geometry of h2o. Definition of electronegativity electronegativity is a measure of how much an atom attracts an electron. Neither atom completely loses or gains electrons as in ionic bonding.
How to tell if a bond is ionic, covalent or polar covalent. What is the main difference between polar molecules and ionic molecules? Ionic bond, covalent bond, james bond, so many bonds!
You have to calculate the difference in electronegativities between the atoms. The higher the electronegativity, the. And we also have to check the molecular geometry of c4h8o2.
Decide whether a bond between the two atoms given is going to be polar, nonpolar or ionic learn with flashcards, games, and more — for free.