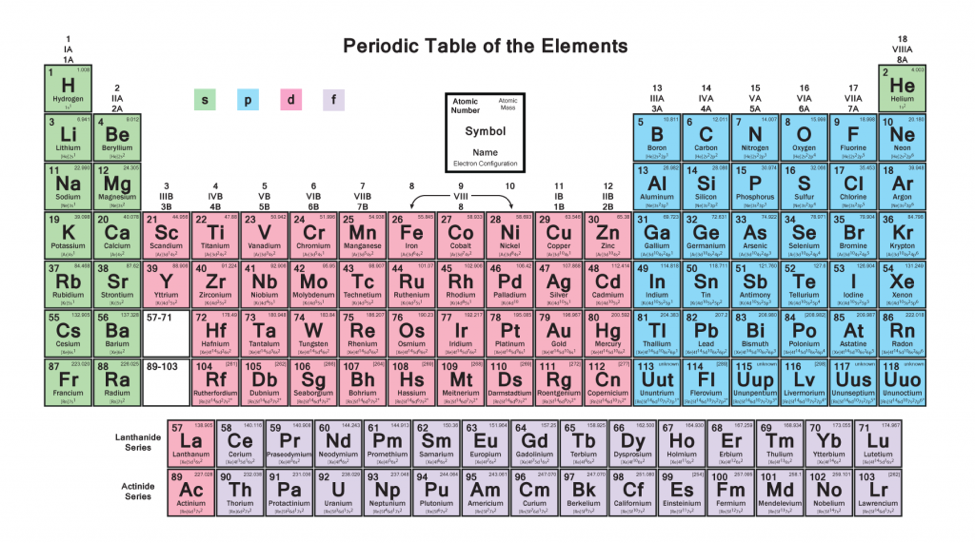
A block of the periodic table is a set of elements unified by the atomic orbitals their valence electrons or vacancies lie in. Fourteen electrons now according to pauli exclusion principle each orbital must have only two electrons thus s subshell has two electrons p subshell has six electrons d subshell has ten electrons and f subshell has fourteen electrons in total.

Block (Periodic Table) - Wikipedia
If you see any periodic table that lists those f elements by omitting the very end, which are lutetium and lawrencium, and incorporating them into the inner transition metals, that depiction is wrong.

How many electronds in f block. Each block is named after its characteristic orbital: The term appears to have been first used by charles janet. P− block − 6 electrons 3.
D− block − 10 electrons 4. F− block − 14 electrons Properties of a fluorine atom.
The highest energy level (valence shell) contains only 1 electron in an s subshell. The labels s, p, d and f blocks of the periodic table refer to the subshell that is being filled with electrons. For an f subshell, it is fourteen electrons.
Not all of the lanthanides have 5d electrons, only la, ce, gd and lu do! The block names (s, p, d, and f) are derived from the spectroscopic notation for the value of an electron's. How many electrons can the f block hold?
This is what wikipedia shows : How many electrons can the f orbital hold. S− block − 2 electrons 2.
Maximum capacity for an s subshell is two electrons. How many protons, neutrons and electrons does fluoride ion(f. The rest fill the 6s.
Those electrons are sitting in the f orbital. These elements have electrons, (1 to 14) in the f orbital, (0 to 1) in the d orbital of the penultimate energy level and in the outermost’s orbital. Electron configuration 2s 2 2p 5:
If so can you give examples? The elements are 4f series of ce to lu and 5f series of th to lw. An element’s location on the periodic table is based on where the last electron to get to the performance is sitting.
For a p subshell, it is six electrons, for a d subshell, it is ten electrons. Medium solution verified by toppr number of electrons in each s−,p−,d− and f− blocks/subshell are as follows: How many number of unpaired electrons are present in mn^2 + ?
There are only 14 elements in the f block by definition. This article discussed in detail how to easily find the number of protons, neutrons, and electrons in a fluorine atom. ⚛ group 1 elements occur at the beginning of a new row (period) of the periodic table.
Chemistry electron configuration electron configuration 2 answers david drayer dec 9, 2016 the f block has the lower level filled, then for valance electrons has 2 s electrons 1 d electron and then up to 14 f electrons filling the 7 f orbitals. How many electrons can be included in the s−,p−,d− and f− blocks? So, for instance, looking at #u#.
There are two series in the f block corresponding to the filling up of 4f and 5f orbitals. But for most of the transition and inner transition elements, the valence electrons are the electrons present in the shells outside the noble gas core. #[rn]7s^2 5f^4# in an anomalous case, which is quite nuanced (which you may not need to know), #u# actually has a different electron configuration than expected:
Cc | Electron Configurations: A Must Know Hack

F Block Orbital Filling And Electron Configuration - Youtube